Ministry of Health, Labour and Welfare
| Target of Generic Share by Volume | 60% or above |
|---|
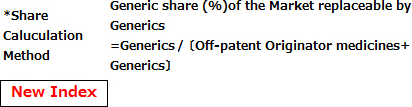
| Target Date | By the End of FY 2017 (End of March,2018) |
|---|
| Scope of Approaches |
|---|
| 1.Stable Supply |
| 2. Ensuring Confidence in Quality |
| 3. Information Provision |
| 4. Improvement of Environment for Promotion of Generic Medicine Use |
| 5. NHI(National Health Insurace) related Matters |
| 6. Monitoring of Implementation Status |
(Reference1) New Index and Old Index of Market share by volume on the basis of MHLW market survey (Sep. 2011)
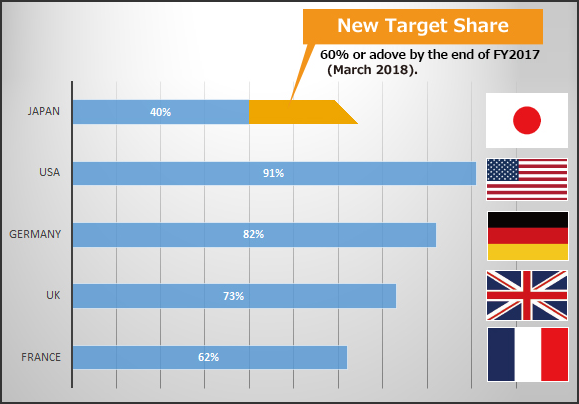
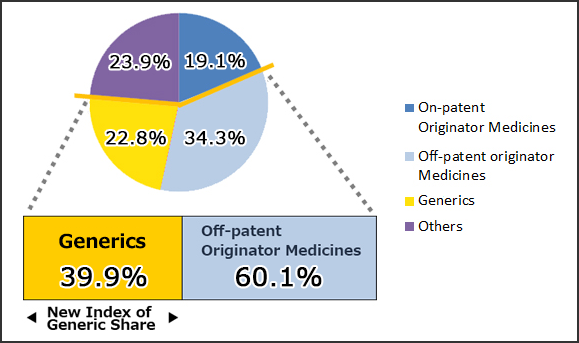
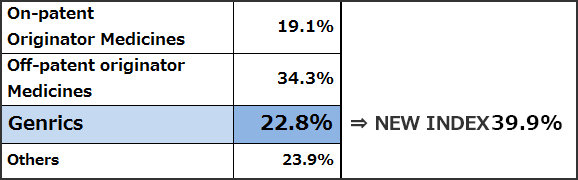
Source : IMS Health, MIDAS Market Segmentation 2010
(Reference 2) Summary of Approaches for Further Promortion of Generic Medicine Use indicated in "The Roadmap 2013"
1. Stable Supply
- Preparation of a guideline for stable supply (Industrial Association)
- Development of a manual based on the guideline (Each Company)
- Development a plan of production capacity and APIs procurement in response to future expansion of generic market (Each Company)
- Providing information on shortage in oversea countries and administrative guidance in problematic cases of stable supply(Central Government)
2. Ensuring Confidence in Quality
- Continuation of survey of scientific literatures on quality of generics, the Review Committee on Quality of Generics and quality monitoring of generics on the market (Central Government)
- Conduct of educational activities so as to get better understanding about quality of generics (Local Government)
- Quick response to the issues identified in the literatures (Each Company)
3. Information Provision
- Improvement and expansion of the existing Generic Medicine Information Servicies operated by industirial association (Industrial Association)
- Improvement of information collection and provision (Each Company)
- Use of community-level council for promotion of generic medicine use (Local Government)
- Preparing a list of widely-used generics in each community(Local Government)
4. Improvement of Environment for Promotion of Generic Medicine Use
- Action in the line with the National Health Care Cost Containment Plan (Central and Local Government)
- Expansion of notification to patients concerning the price difference between generics and originator medicines (Insurance Bodies)
5. NHI(National Health Insurance) related Matters
Consideration by the Central Social Insurance Medical Council etc., (Central Government)
6. Monitoring of Implementation Status
Conduct of monitoring on implentation status of approaches and evaluation of its results (Central Government)